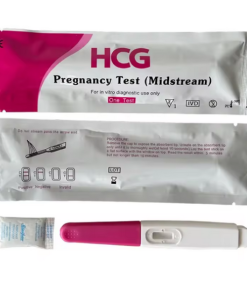
Ecotest Human Chorionic Gonadotropin Pregnancy Test Kit
The Ecotest Human Chorionic Gonadotropin (hCG) Pregnancy Test Kit is a rapid, qualitative immunoassay for professional use that can detect pregnancy from either a urine or serum sample. It provides highly accurate results by identifying the presence of the hCG hormone, a key marker of pregnancy, in minutes.
SKU: BL-RapidDTK-05
Category: Rapid Diagnostics Kits
Key Features and Benefits
- Dual Specimen Compatibility: The primary advantage is its ability to use either a urine sample (preferred for point-of-care or early morning use) or a serum/plasma blood sample (collected via standard clinical procedure).
- High Accuracy & Sensitivity: The test boasts an accuracy and specificity of over 99.9%. The sensitivity level is typically around 25 mIU/mL for standard kits, although some enhanced sensitivity versions can detect levels as low as 10 mIU/mL.
- Rapid Results: Results are typically available within 3 to 5 minutes.
- Easy to Use: The test uses a simple cassette format where a few drops of the specimen are added to a sample well, with clear visual lines indicating the result.
- Internal Control: Each test includes a control line (“C” region) that confirms the test is working correctly and sufficient specimen volume has been used.
- Long Shelf Life: The kits can be stored at room temperature or refrigerated (2-30°C) and are stable until the expiration date printed on the sealed pouch.
Procedure for Use (Professional)
- Specimen Collection: Collect urine in a clean, dry container or a serum/plasma blood sample following standard clinical procedures. Ensure the specimens are at room temperature before testing.
- Testing: Remove the test cassette from the sealed pouch. Using a dropper, transfer 3 full drops of the specimen (urine or serum) into the specimen well(s) of the cassette.
- Reading Results: Wait for the colored lines to appear. Interpret the results at 3-5 minutes. Do not interpret the results after the maximum read time (typically 10 minutes).
- Positive Result: Two distinct colored lines appear (Control “C” and Test “T”). This indicates the presence of hCG.
- Negative Result: Only one colored line appears in the control region “C”.
- Invalid Result: The control line fails to appear. The test is invalid and should be repeated with a new device.
A confirmed pregnancy diagnosis should only be made by a physician after all clinical and laboratory findings have been evaluated.
Related products
Rapid Diagnostics Kits
Rapid Diagnostics Kits
Rapid Diagnostics Kits
Rapid Diagnostics Kits
Rapid Diagnostics Kits